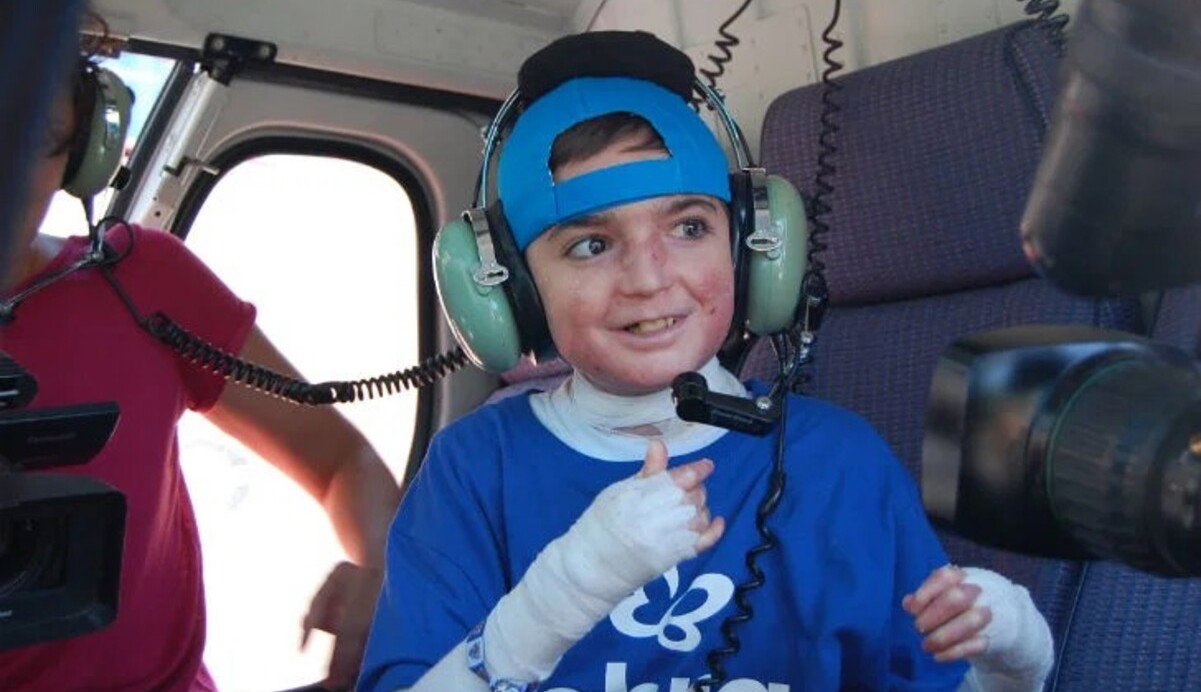
For the first time in Spain, two people living with dystrophic epidermolysis bullosa, commonly known as butterfly skin, have begun receiving a dedicated gene therapy. Adrian, a 22-year-old from Tolox in Malaga and Leo, a 12-year-old boy from Seville are receiving medical treatment at the public hospitals Costa del Sol in Marbella and Virgen del Rocio in Seville.
What is butterfly skin and how does Vyjuvek work?
Dystrophic epidermolysis bullosa is a rare, incurable genetic condition that causes extreme fragility of the skin and some mucous membranes. Even a minor touch, a light knock, or tight clothing can trigger blisters and wounds resembling third-degree burns. Daily wound care, cleaning lesions, applying specialist dressings, managing pain, and monitoring for infection typically takes between two and five hours every day.
Vyjuvek is the first drug developed specifically for this condition. Applied directly to wounds and lesions, it works by delivering a healthy copy of the faulty gene into skin cells, helping the body produce a protein it cannot make on its own, one that essentially acts as a glue keeping the skin’s layers bonded together.
Andalucia leads the way
Andalucia has become the first autonomous community in Spain to administer Vyjuvek within its public health system. Regional president Juanma Moreno described it as “the great day has arrived,” confirming that the Andalusian Health Service is currently covering the full cost of treatment whilst national funding negotiations continue. At approximately €100,000 per patient per month, the accumulated cost over a patient’s lifetime could reach several million euros, which explains the complexity of securing permanent state funding. Around 45 people in Andalusia are thought to be eligible for the therapy.
The stories of the boys
Adrian studies at the University of Malaga and is accompanied daily by his mother in case of emergencies. Over his lifetime he has undergone a gastrostomy, multiple hand surgeries to prevent loss of his fingers, and a corneal transplant. He has spent years managing pain and hours of daily wound care.
Leo had been “waiting for this moment his whole life,” as he explained himself on the day of his first treatment, adding he felt “nervous, but ready.” His mother Lidia said the therapy would “change their lives,” whilst urging that it reach all who need it across Spain.
Campaigns by families and DEBRA
Behind this incredible development lies years of sustained pressure from patient organisation DEBRA Piel de Mariposa. A campaign they launched in October 2025 gathered more than 150,000 signatures, which were delivered to the Health Minister to support the inclusion of Vyjuvek in Spain’s public health provision. In February 2026, two affected families, including Leo’s, took their case to the European Parliament.
DEBRA director Evanina Morcillo welcomed the first cases as “a hopeful step, but not sufficient,” warning that access to a life-changing therapy must not depend on a patient’s postcode. National inclusion in Spain’s health service catalogue remains the goal for all affected families.
For more information on this rare disease and the charity visit the DEBRA official website.

 Caso Koldo3 weeks ago
Caso Koldo3 weeks ago
 New Developments4 weeks ago
New Developments4 weeks ago
 New Developments4 weeks ago
New Developments4 weeks ago
 Ataque contra Irán4 weeks ago
Ataque contra Irán4 weeks ago
 New Developments4 weeks ago
New Developments4 weeks ago